Posted on Feb 3, 2023
Manufacturer recalls eye drops after possible link to bacterial infections
465
9
3
5
5
0
Posted >1 y ago
Responses: 3
PO1 William "Chip" Nagel
..."Patients reported using more than 10 different brands of artificial tears between them, but the EzriCare product, which is sold over the counter and is preservative-free, was mentioned consistently, the CDC said.
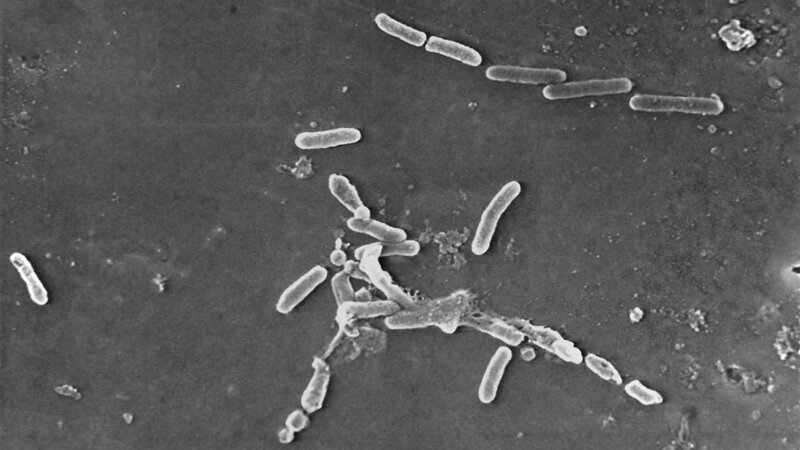
The CDC said an outbreak of this particular strain of Pseudomonas aeruginosa, VIM-GES-CRPA, has not been detected in the U.S. before.
Opened bottles of EzriCare artificial tears matched the outbreak strain. The CDC is currently testing unopened bottles of the product to see if the bottles became contaminated during manufacturing. It is recommending that clinics and patients pause their use of the product until both the CDC and FDA issue additional guidance.
Those who have used the product may begin showing symptoms of an eye infection, which include yellow, green, or clear discharge from the eye, eye pain or discomfort, redness of the eye or eyelid or blurry vision."
..."Patients reported using more than 10 different brands of artificial tears between them, but the EzriCare product, which is sold over the counter and is preservative-free, was mentioned consistently, the CDC said.
The CDC said an outbreak of this particular strain of Pseudomonas aeruginosa, VIM-GES-CRPA, has not been detected in the U.S. before.
Opened bottles of EzriCare artificial tears matched the outbreak strain. The CDC is currently testing unopened bottles of the product to see if the bottles became contaminated during manufacturing. It is recommending that clinics and patients pause their use of the product until both the CDC and FDA issue additional guidance.
Those who have used the product may begin showing symptoms of an eye infection, which include yellow, green, or clear discharge from the eye, eye pain or discomfort, redness of the eye or eyelid or blurry vision."
(1)
(0)
Read This Next

 Medical
Medical Health
Health Drugs
Drugs Pharmaceuticals
Pharmaceuticals