Posted on Apr 20, 2022
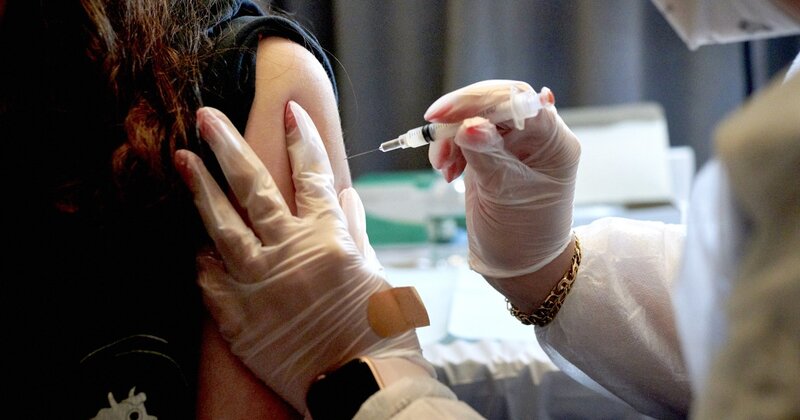
Moderna says redesigned Covid booster provides better protection
586
19
2
10
10
0
Posted 2 y ago
Responses: 2
PO1 William "Chip" Nagel
..."For certain indications, the Moderna COVID‑19 Vaccine has not been approved or licensed by the US Food and Drug Administration (FDA), but has been authorized for emergency use by the FDA, under an Emergency Use Authorization (EUA), to prevent Coronavirus Disease 2019 (COVID‑19) in individuals 18 years of age and older. There are two presentations/formulations of the Moderna COVID‑19 Vaccine authorized for use."...
..."For certain indications, the Moderna COVID‑19 Vaccine has not been approved or licensed by the US Food and Drug Administration (FDA), but has been authorized for emergency use by the FDA, under an Emergency Use Authorization (EUA), to prevent Coronavirus Disease 2019 (COVID‑19) in individuals 18 years of age and older. There are two presentations/formulations of the Moderna COVID‑19 Vaccine authorized for use."...
(0)
(0)
Read This Next

 Medical
Medical Health
Health Drugs
Drugs Pharmaceuticals
Pharmaceuticals