https://www.npr.org/sections/health-shots/2022/04/26/ [login to see] /pfizer-asks-fda-to-authorize-booster-shots-for-kids-ages-5-through-11
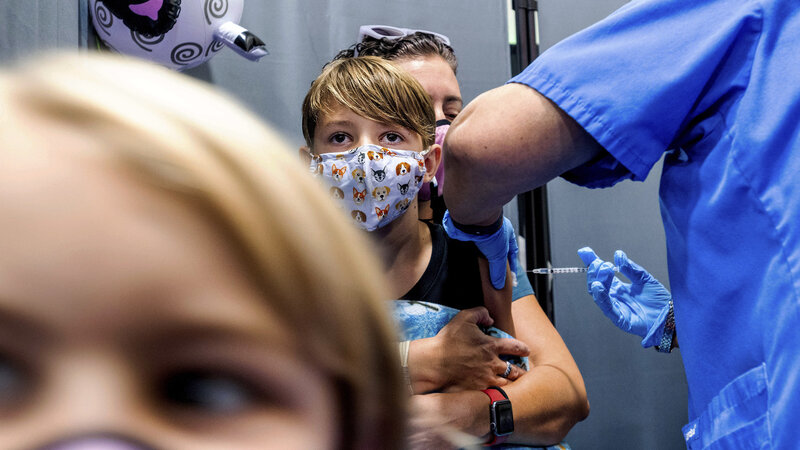
Children ages 5 through 11 who've received two shots of the Pfizer COVID-19 vaccine may soon be eligible for a booster. That's if the Food and Drug Administration agrees to a request made Tuesday by pharmaceutical companies Pfizer and BioNTech to authorize their booster shot for kids in this age range.
The companies submitted data to the FDA showing that the low-dose booster shot is safe for children ages 5 through 11 and could help protect them against omicron. Currently, boosters are only authorized for people ages 12 and older.
The companies say a third shot for younger kids could help counter waning immunity that has left other age groups more vulnerable, especially against omicron, which is better at getting around the immune system.
Some independent experts agree. But others remain skeptical that another shot is needed yet for younger kids.
"Over time, immunity to the vaccine wanes. And we know that that happens in children as well. So the data right now really support vaccine and boosters in particular to maintain immunity," says Dr. Yvonne Maldonado, an infectious disease researcher at Stanford who advises the American Academy of Pediatrics and also has been helping test the vaccine for Pfizer.

 Children
Children Medical
Medical Health
Health Drugs
Drugs Pharmaceuticals
Pharmaceuticals